
I want to summarise the results of this before looking at specific examples in detail. It isn't essential for following the rest of the current page. This is the second page in a series of pages about redox potentials, and you will probably need to read the first page as well. If you want to read more about the electrochemical series, including the origin of these numbers, you will find it by following this link. Note: For the purposes of electrolysis, you don't need to understand where these numbers come from, or what exactly the equilibria apply to. The lower down the electrochemical series something on the left-hand side of the equilibrium is, the more readily it will pick up electrons. The higher up the electrochemical series something on the right-hand side of the equilibrium is, the more readily it will lose electrons. The electrochemical series can be thought of as an extended, and slightly modified, reactivity series.Īll you really need to know as far as electrolysis is concerned is: It won't be easy to remove electrons to make gold ions, but it will be easy to convert gold ions back into gold metal again. So gold won't be very reactive, because it has a very positive E° value. That also means that something like lithium will have little tendency to pick up electrons to form atoms once it has ionised.īy contrast, something with a positive E° value will be reluctant to lose electrons to form ions, but it will be quite easy to make one of its ions pick up electrons to make the neutral element again. That means that the more negative the E° value, the greater the tendency for one of these elements to lose electrons and form their ions. The more negative the E° value (usually read as "E-nought"), the further to the left the position of equilibrium lies. The table below lists a few metals (and hydrogen) showing their tendency to lose electrons. That means that you may have more than one ion arriving at each electrode, and there can be a choice over which gets discharged.įor example, if you electrolysed sodium chloride solution, sodium ions and hydrogen ions (from the water) are both attracted to the cathode, and chloride ions and hydroxide ions (from the water) are both attracted to the anode You should know that a hydrogen ion doesn't exist on its own in these circumstances - it actually joins to another water molecule to give a hydroxonium ion, H 3O +. 
Water itself is a very weak electrolyte, because it splits to a very small extent into hydrogen ions and hydroxide ions. The situation is more complicated when you electrolyse a solution rather than a melt because of the presence of the water. The role of water in the electrolysis of aqueous solutions of electrolytes Most people will have met quite a lot of this in chemistry courses for 14 - 16 year olds. It is physically impossible to measure the potential of a single electrode: only the difference between the potentials of two electrodes can be measured (this is analogous to measuring absolute enthalpies or free energies recall that only differences in enthalpy and free energy can be measured.) We can, however, compare the standard cell potentials for two different galvanic cells that have one kind of electrode in common.This page looks at the electrolysis of aqueous solutions of compounds. Measured redox potentials depend on the potential energy of valence electrons, the concentrations of the species in the reaction, and the temperature of the system. 1 atm for gases, pure solids or pure liquids for other substances) and at a fixed temperature, usually 25☌. Corrections for non ideal behavior are important for precise quantitative work but not for the more qualitative approach that we are taking here. To do this, chemists use the standard cell potential (E° cell), defined as the potential of a cell measured under standard conditions-that is, with all species in their standard states (1 M for solutions, concentrated solutions of salts (about 1 M) generally do not exhibit ideal behavior, and the actual standard state corresponds to an activity of 1 rather than a concentration of 1 M. 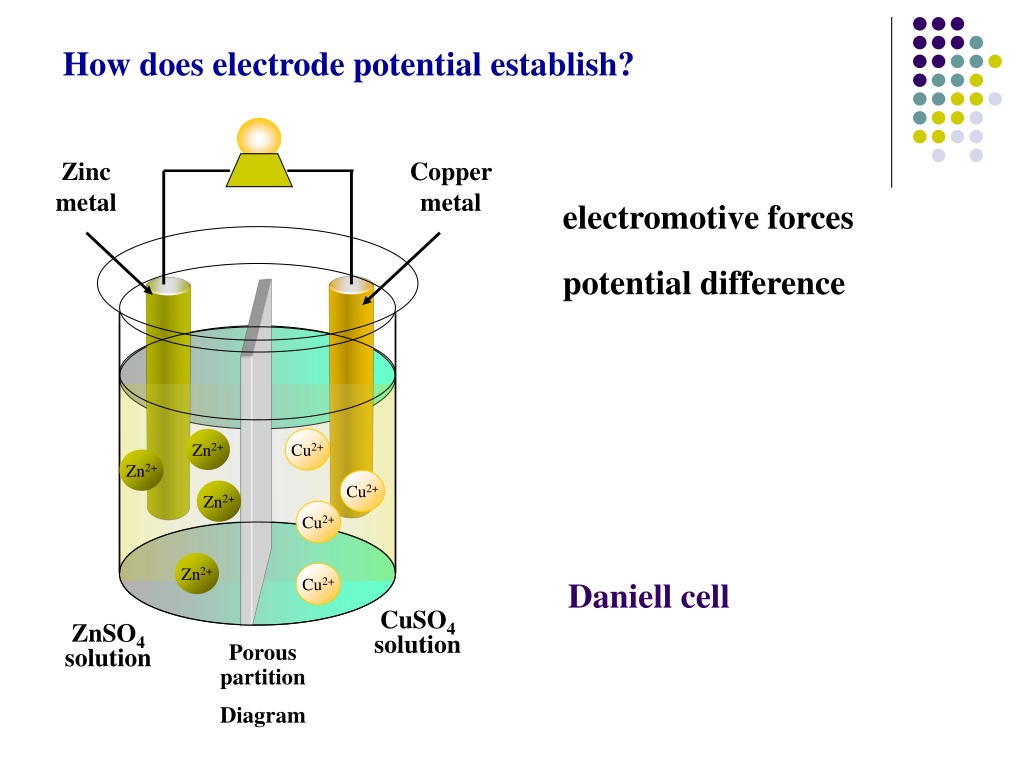
To develop a scale of relative potentials that will allow us to predict the direction of an electrochemical reaction and the magnitude of the driving force for the reaction, the potentials for oxidations and reductions of different substances must be measured under comparable conditions. The measured potential of a cell also depends strongly on the concentrations of the reacting species and the temperature of the system. Thus we can conclude that the difference in potential energy between the valence electrons of cobalt and zinc is less than the difference between the valence electrons of copper and zinc by 0.59 V. \) but instead of copper use a strip of cobalt metal and 1 M Co 2 + in the cathode compartment, the measured voltage is not 1.10 V but 0.51 V.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
